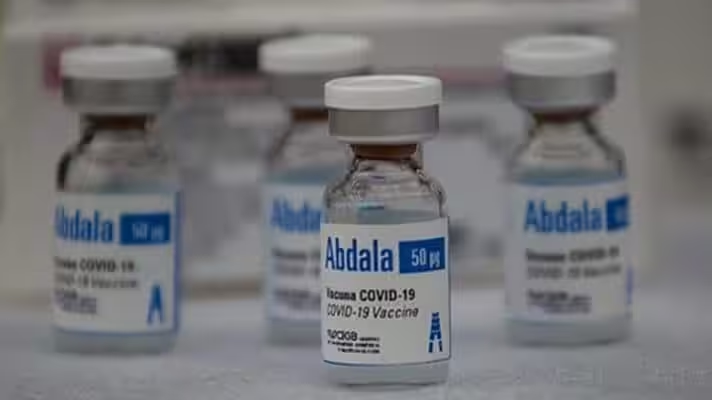
Cuban COVID-19 vaccine Abdala has shown 92.28% effectiveness in clinical trials carried out by the Havana-based Center for Genetic Engineering and Biotechnology (CIGB).
Abdala is one of Cuba’s five COVID-19 vaccine candidates, and the first to be developed in Latin America that functions through a three dose administration cycle. It is in the final stages of clinical trials and is soon expected to receive official approval from the Cuban health authorities.
RELATED CONTENT: Cuba’s COVID-19 Vaccines: A Journey of Collaboration and Revolutionary Solidarity
Cuban President Miguel Mario Díaz-Canel Bermúdez, commenting on the news of Abdala’s high effectiveness rate wrote on Twitter: ”Hit by two pandemics (COVID-19 and the US blockade), our scientists from the Finlay Institute and CIGB have overcome all obstacles and have given us two very effective vaccines.”
The government of Venezuela has established a cooperation agreement with its Cuban counterpart to produce Abdala vaccines at the ESPROMED Bio plant, located in the University City of Caracas.
RELATED CONTENT: Disease as a Weapon: Has the US Blocked Vaccines For Venezuela?
Earlier, on June 19, the Finlay Institute of Vaccines announced that its vaccine candidate Soberana 02 had shown a 62% efficacy after the injection of two of its stipulated three doses. Hence, Cuba has now achieved the goal of having two of its five vaccine candidates meet the minimum efficacy requirements stipulated by the World Health Organization (WHO).
Already 4 million 885 thousand 270 doses of Soberana 02 and Abdala have been administered in Cuba. Nearly 2.25 million Cubans have received at least one dose and more than 905,000 have completed the entire immunization cycle. According to official data, COVID-19 cases have decreased by half in the Cuban capital, Havana, since the vaccination campaign began a month ago.
Featured image: Cuban COVID-19 vaccine Abdala has shown an efficacy rate of 92.28% in clinical trials. Photo: Cubadebate

Misión Verdad
Misión Verdad is a Venezuelan investigative journalism website with a socialist perspective in defense of the Bolivarian Revolution
- Misión Verdad
- Misión Verdad